Safety
WINLEVI IS GENERALLY WELL TOLERATED2,13,14,17


No interaction clinical studies, including interaction studies with other topical treatments, have been performed. Caution should be exercised in using WINLEVI® with other drugs known to suppress the HPA axis.
Please read the safety section and special warnings and precautions for use before prescribing.
Educational materials:
Educational materials regarding these precautions are available for healthcare professionals and patients (or parents/caregivers). A patient card is provided with the package of this medicinal product.
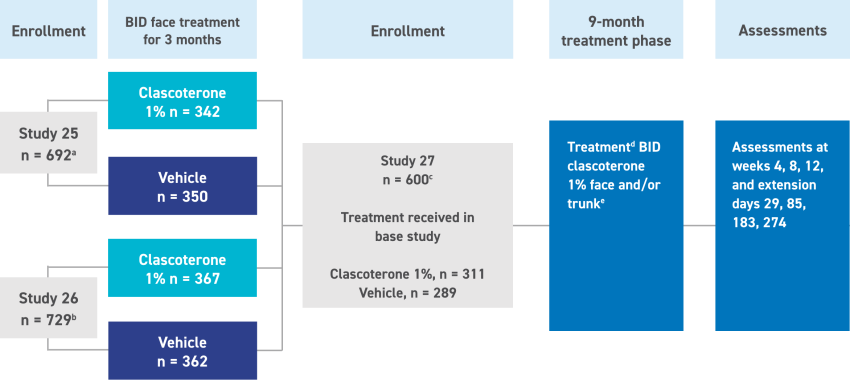
a Number of ITT patients ≥ 12 years of age enrolled in Study 25.
b Number of ITT patients ≥ 12 years of age enrolled in Study 26.
c Number of ITT patients ≥ 12 years of age enrolled in the long-term extension study (Study 27).
d Patients who achieved IGA score of ≤1 could stop treatment and resume if/when acne worsened.
e Total clascoterone treatment duration was up to 12 months for patients treated with clascoterone for 3 months in the pivotal studies.
BID, twice daily.
(adapted from Eichenfield et al. 202417)
TEAE, treatment emergent adverse event.
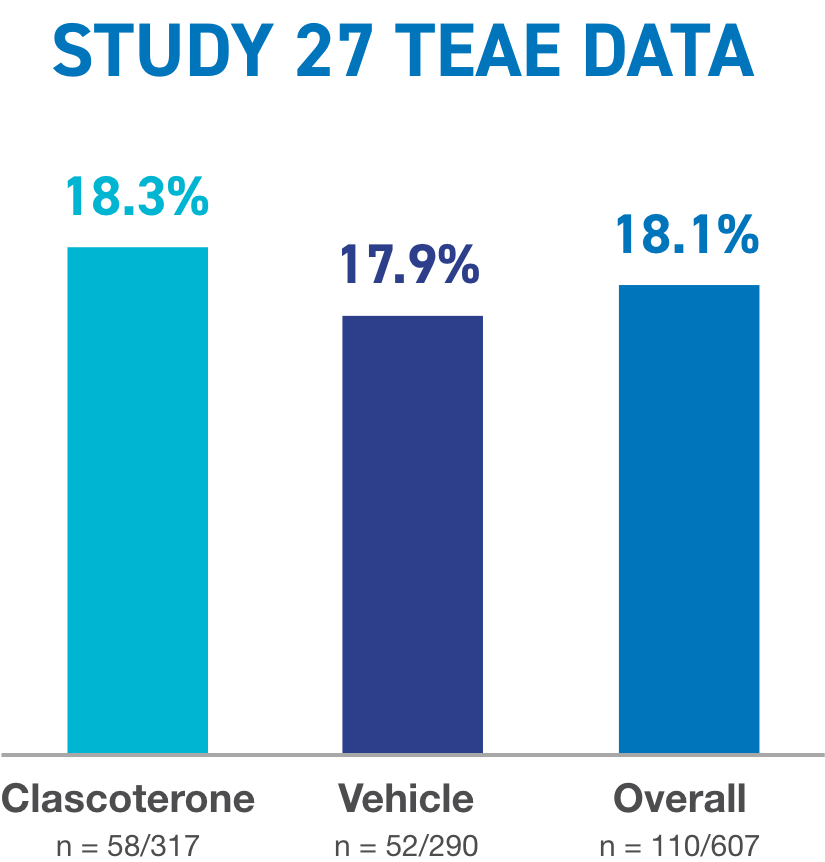
(adapted from Eichenfield et al. 202018)
Among the 444 subjects aged 12 to < 18 years enrolled in phase 2 and phase 3 vehicle-controlled studies for acne vulgaris and exposed to clascoterone cream, the overall incidence of adverse reactions was 4/444 (0.9%). Frequency, type and severity of adverse reactions through week 12 were similar to those in adults as presented in table shown above, which covers both populations.
There are no data on the effect of clascoterone on human fertility. Results from animal studies following subcutaneous administration showed no effect on fertility in male or female rats.
Women of childbearing potential have to use an effective method of contraception during treatment and for at least 10 days after the last dose. No interaction clinical studies have been performed, therefore, an interaction with hormonal contraception cannot be excluded. The pregnancy status should be verified before initiating treatment with clascoterone in women of childbearing potential.
There are no or a limited amount of data from the use of cutaneous clascoterone in pregnant women.
Animal studies have shown reproductive toxicity following subcutaneous administration. Although systemic absorption of cutaneous clascoterone and its main metabolite cortexolone, is negligible, there could be individual factors (e.g. use over large surfaces, prolonged use) that may contribute to an increased systemic exposure. Based on animals studies and its mechanism of action (androgen receptor inhibition), clascoterone can cause fetal harm.
This medicinal product is contraindicated during pregnancy.
The patient must be informed and understand the risks related to the use of this medicinal product during pregnancy.
It is unknown whether clascoterone/metabolites are excreted in human milk. A risk to the newborns/infants cannot be excluded.
Use of this medicinal product is not recommended while breast-feeding or breast-feeding should be discontinued during treatment with this medicinal product.
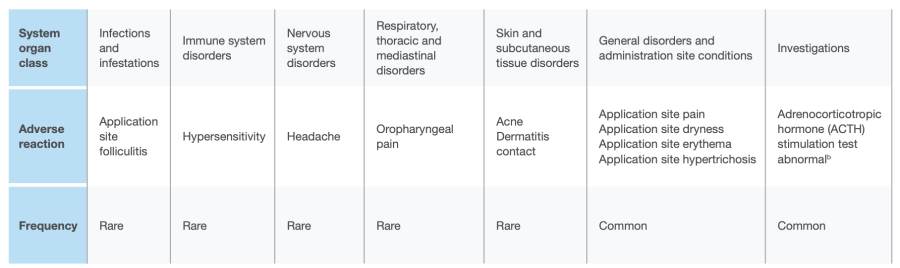
a Frequencies have been evaluated according to the following convention: very common (≥ 1/10), common (z 1/100 to <1/10), uncommon (2 1/10,000 to <1/1,000), very rare (<1/10,000) and not known (cannot be estimated from the available data).
b Assessed in the dedicated phase 2 study at supratherapeutic dosages.

WINLEVI® (clascoterone) 10 mg/g cream is indicated for the topical treatment of acne vulgaris in patients 12 years of age and older.3
Adverse events should be reported. Reporting forms and information can be found at yellowcard.mhra.gov.uk. Adverse events should also be reported to Glenmark Pharmaceuticals Europe Ltd medical_information@glenmarkpharma.com or call 0800 458 0383.
This medicine is subject to additional monitoring. This will allow quick identification of new safety information. You can help by reporting any side effects you may get. See yellowcard.mhra.gov.uk for how to report side effects.
Depicted people are not actual patients.