Clinical evidence
Proven Efficacy, Consistent across trials

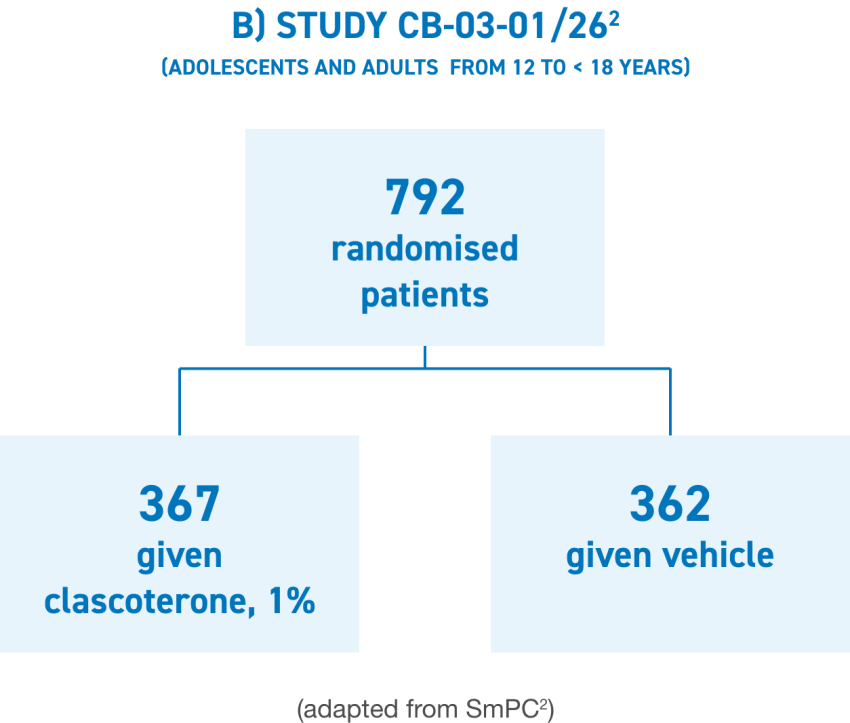
WINLEVI® was studied in 2 identical phase III pivotal trials and was statistically superior over vehicle for all coprimary endpoints.2


WINLEVI® was studied in 2 identical phase III pivotal trials and was statistically superior over vehicle for all coprimary endpoints.2


IGA, Investigator’s Global Assesment; ILC, inflammatory lesion count; NILC, noniflammatory lesion count; TLC, total lesion count.

SIGNIFICANTLY INCREASED
IGA TREATMENT SUCCESS2,b
a The vehicle cream was identical to clascoterone 10 mg/g cream, in terms of colour, consistency, and smell, and the creams were packed in identical blinded tubes.14 The difference between placebo and vehicle is that the vehicle is equivalent to active drug, minus the active component. Thus, vehicle contains only relatively inert substances, while a placebo is intended to have no pharmacological activity. 15
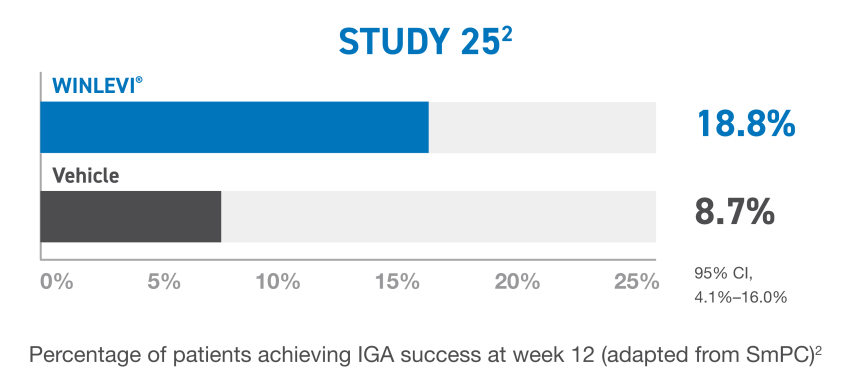
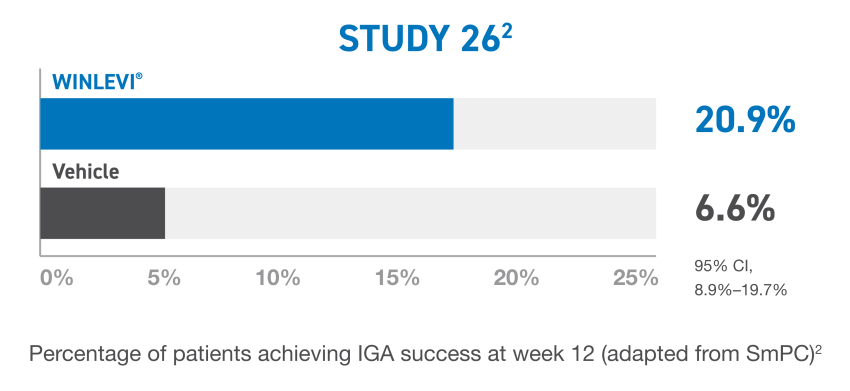
b IGA success was defined as at least a 2-point reduction in IGA score from baseline and an IGA score of 0 (clear) or 1 (almost clear).2
CI, confidence interval; IGA, Investigator’s Global Assesment.
Treatment success was defined as an IGA score of 0 (clear) or 1 (almost clear), and a 2-grade or greater improvement from baseline and absolute change from baseline in NILC and ILC counts at week 12.14
Safety measures included adverse event frequency and severity.14

(adapted from Hebert et al. 202016)
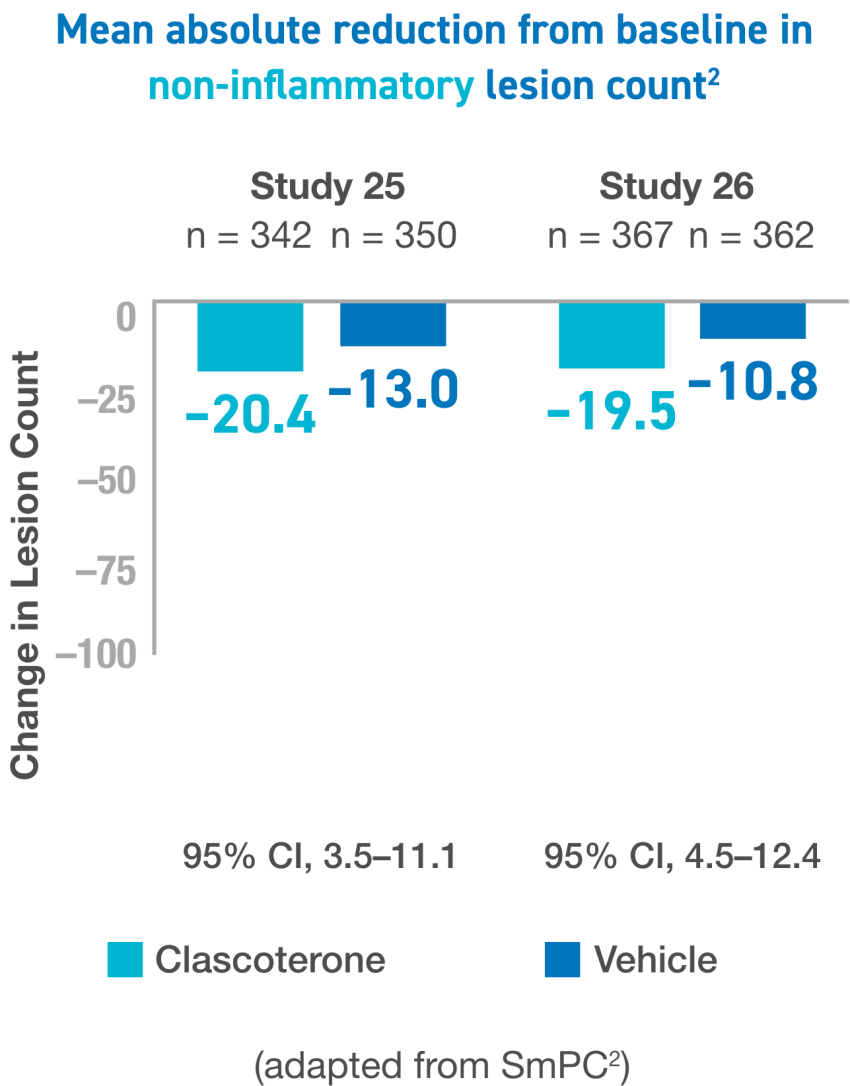
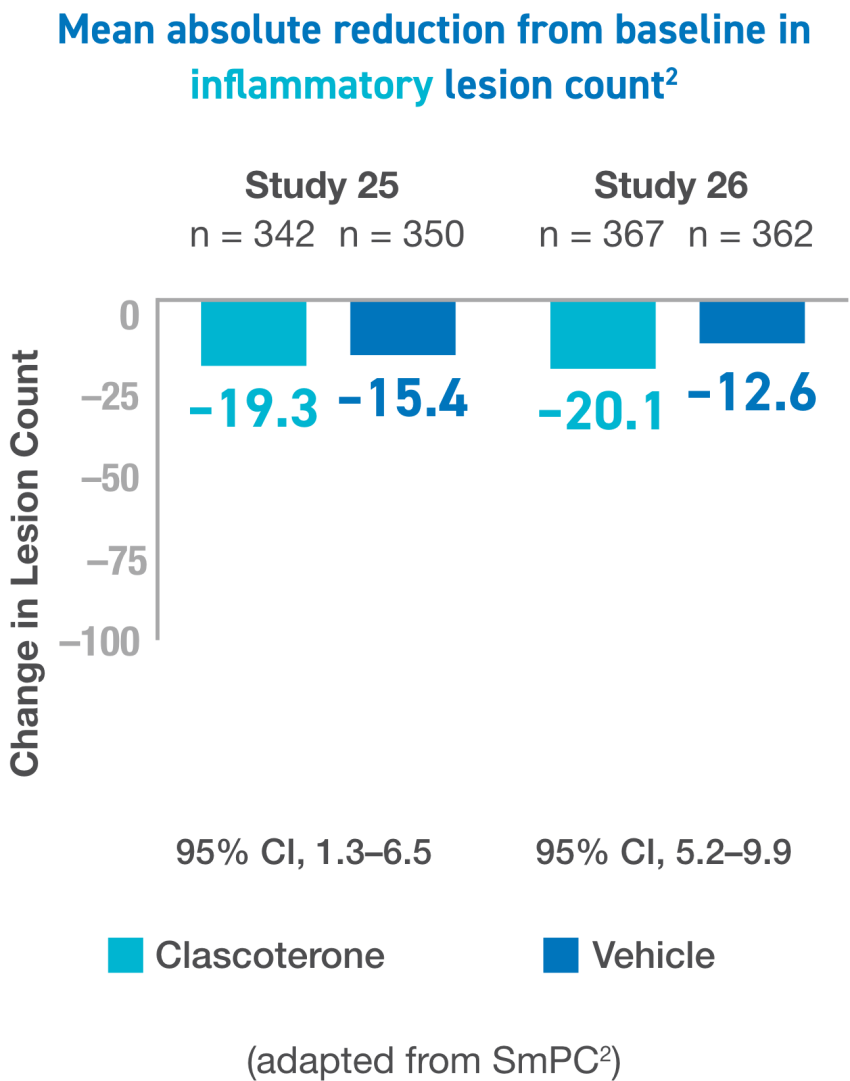
IGA, Investigator’s Global Assesment; ILC, inflammatory lesion count; NILC, noniflammatory lesion count.
a Adjusted proportion of patients with treatment success defined as at least 2-point reduction in IGA vs baseline and an IGA score of 0 or 1 at week 12.
CI, confidence interval; IGA, Investigator Global Assessment; ITT, intention to treat.
For the 3 primary endpoints, proportion of subjects aged ≥12 years achieving “success” at week 12, absolute change from baseline in NILC at week 12, and absolute change from baseline in ILC at week 12, the results were similar between the 2 studies and, as expected based on the significant results in each study.2

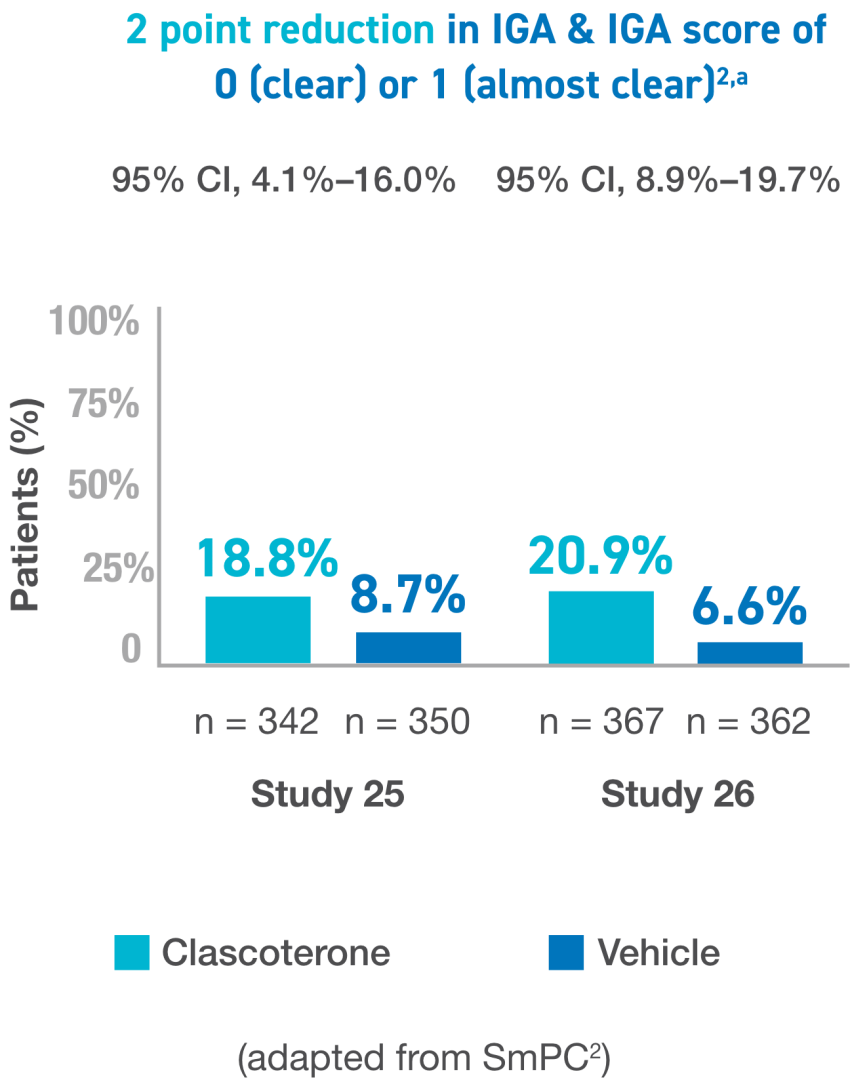
a Investigator Global Assessment (IGA) success was defined as at least a 2-point reduction in IGA compared to baseline and an IGA score of 0 (clear) or 1 (almost clear).
CI, confidence interval; IGA, Investigator’s Global Assesment; ILC, inflammatory lesion count; NILC, noniflammatory lesion count.
Secondary efficacy end points for both studies included absolute and % change from baseline in TLC at week 12. The percentage change from baseline to week 12 was statistically significantly greater in the clascoterone group than the vehicle group for TLC.14,17
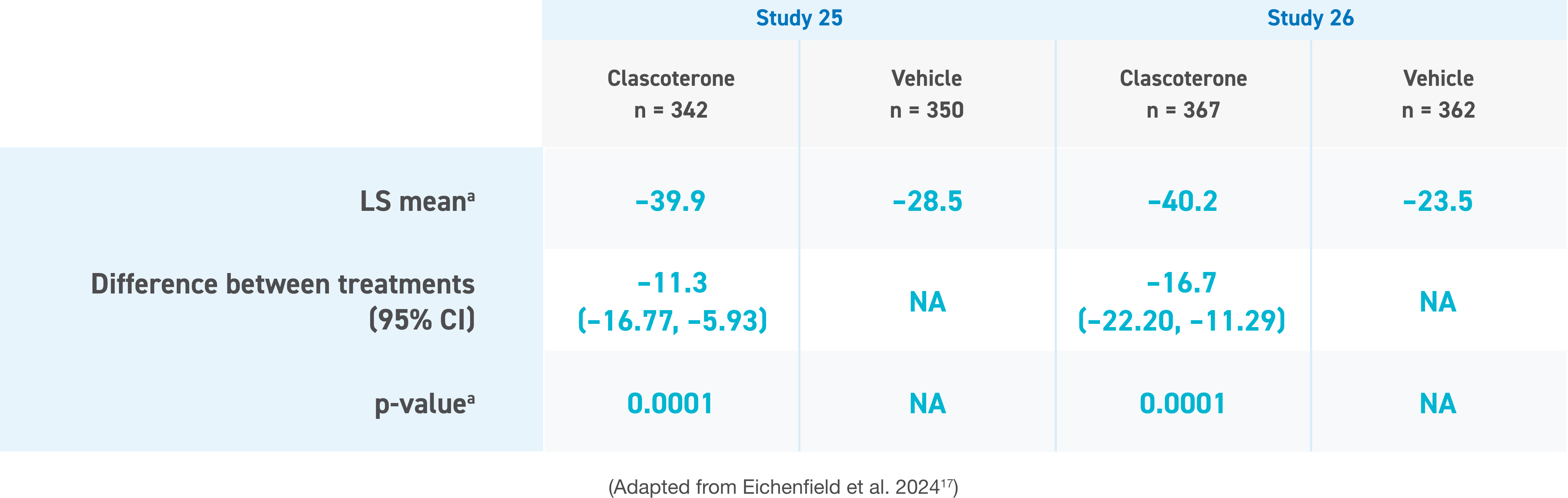
a LS mean and p-value are based on analysis of covariance with treatment and analysis center as fixed effects, and baseline value as a covariate.
CI, confidence interval; LS, least square; NA, not applicable; TLC, total lesion count.

WINLEVI® (clascoterone) 10 mg/g cream is indicated for the topical treatment of acne vulgaris in patients 12 years of age and older.3
Adverse events should be reported. Reporting forms and information can be found at yellowcard.mhra.gov.uk. Adverse events should also be reported to Glenmark Pharmaceuticals Europe Ltd medical_information@glenmarkpharma.com or call 0800 458 0383.
This medicine is subject to additional monitoring. This will allow quick identification of new safety information. You can help by reporting any side effects you may get. See yellowcard.mhra.gov.uk for how to report side effects.
Depicted people are not actual patients.